
U.S. sanctions on Chinese companies operating in Xinjiang or with the Chinese military have reshaped supply chains from agriculture to technology. It’s a different story for the pharmaceutical industry.
For multinational drugmakers with R&D investments and human trials running at Chinese hospitals, there is no regulation prohibiting drug development or clinical testing in a region otherwise subject to a range of existing U.S. sanctions and restrictions.
Data put together by The Wire China and WireScreen from ClinicalTrials.gov, a medical trial database maintained by the U.S. National Library of Medicine, shows that multinational companies have for years actively been sponsoring medical trials taking place in Xinjiang and at People’s Liberation Army-affiliated sites.
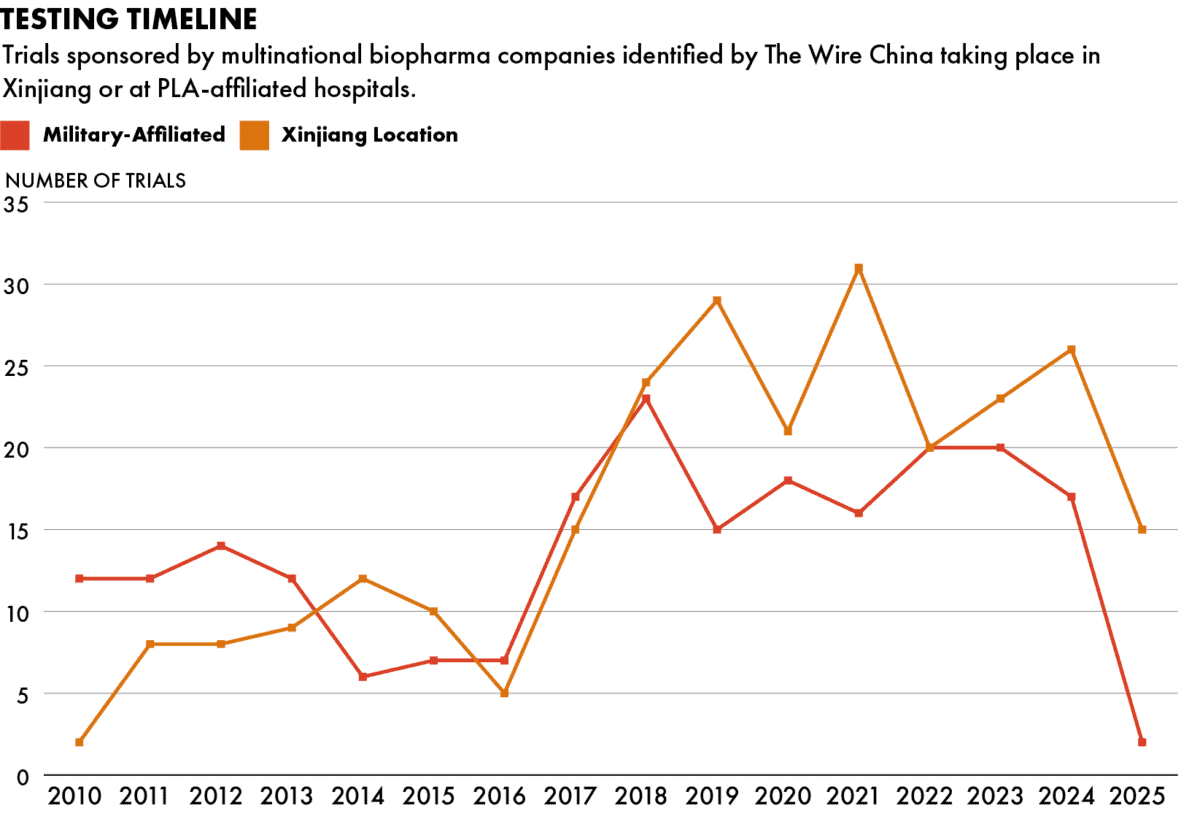
The true scope of this testing is difficult to measure. ClinicalTrials.gov requires registered trials to conform to regulations from regional authorities, but some trials only list the city where the test is held, not the hospital. PLA hospitals also do not always openly identify their military affiliation. China has 165 PLA hospitals across each of its provinces, according to WireScreen data.

For multinational pharmaceutical firms, these sites may just be one of dozens of worldwide locations testing a drug. Testing at multiple locations allows companies to collect data on how medicine impacts a variety of people under unique conditions. The results are used by companies seeking approval from regulatory agencies to market the final product.
One Covid-era treatment, developed by UK-headquartered AstraZeneca, illustrates the role Xinjiang played in the testing of a medicine that received regulatory approval in the United States. The treatment, which was eventually sold under the name Evusheld, was trialed at a hospital in Xinjiang and at other sites across China, the United States, Europe, Latin America, and Japan.
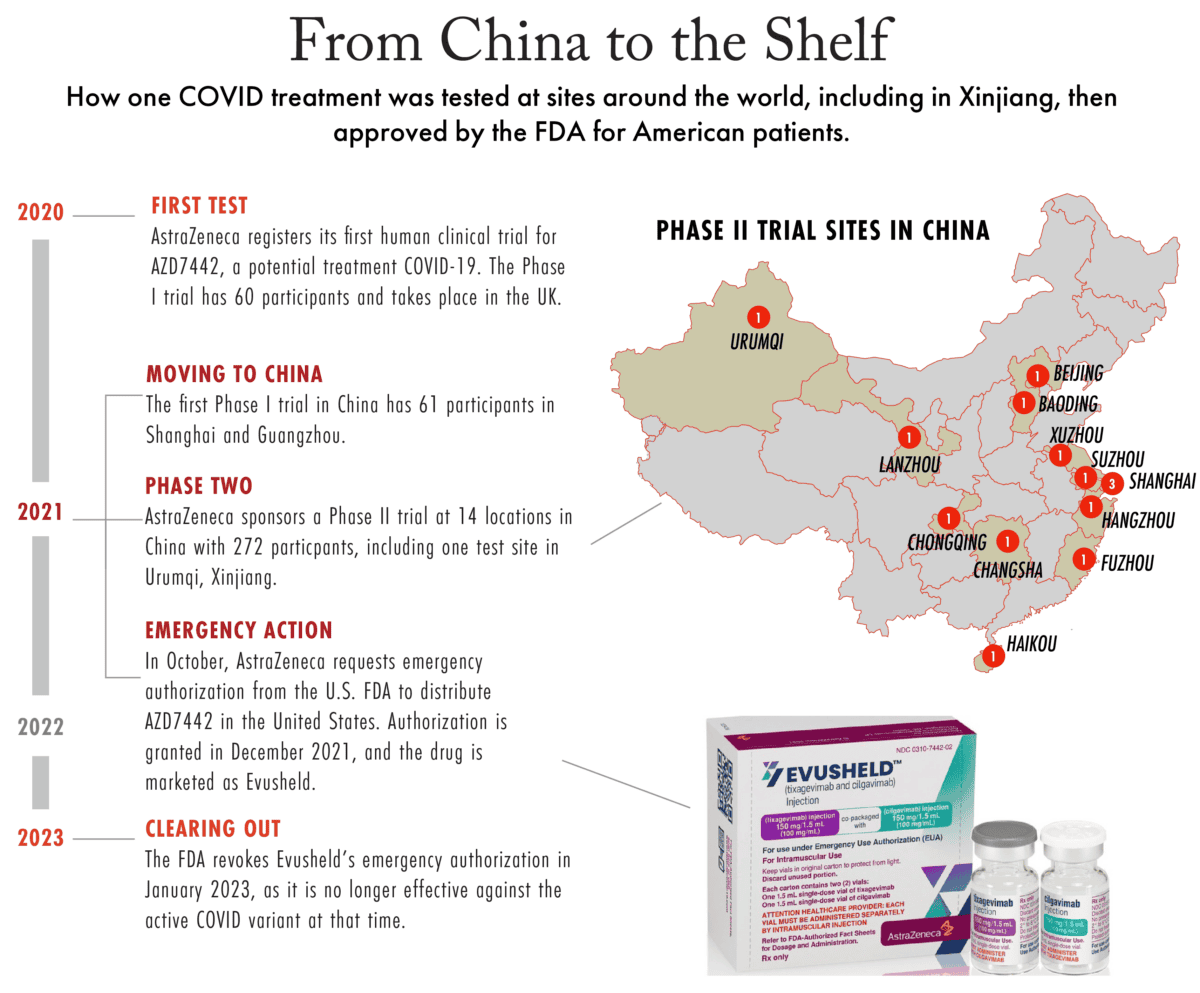
Evusheld’s Xinjiang clinical trial was just one of hundreds sponsored by AstraZeneca between 2021 and 2023. Yet its example illustrates how the pharmaceutical industry can operate in Xinjiang and bring products to market in the United States without facing regulatory requirements addressing forced labor concerns in the region. Several foreign governments and the United Nations have said human rights violations are rife in Xinjiang.

The pharmaceutical industry’s exemption from Xinjiang-related restrictions is not new. Unlike agricultural and technology firms operating in the region, drug manufacturers were exempt from sanctions under the Uyghur Forced Labor Prevention Act, according to a 2024 report from the Washington-based research group Center for Advanced Defense Studies. The act, passed in 2021, prevents the import of all goods mined, produced, or manufactured in Xinjiang unless a forced labor-free supply chain is otherwise proven.
As a result, the industry has been able to continue testing medicines on patients in Xinjiang and at military hospitals. Data identified by The Wire shows the top multinational sponsors of trials over the past fifteen years.
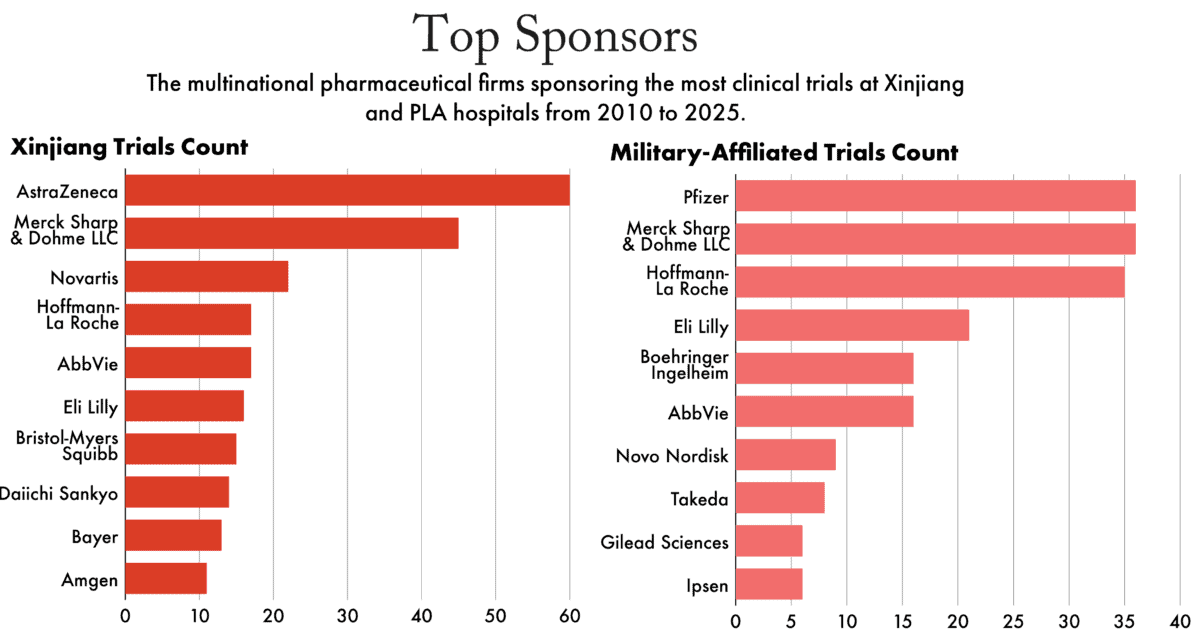
The lack of oversight prompted the House of Representatives’ bipartisan Select Committee on the Chinese Communist Party to address the Food and Drug Administration (FDA) in 2024 and call for more information on pharmaceutical trials conducted in Xinjiang and at PLA hospitals.
“China runs trials at PLA military hospitals and in Xinjiang, where there has been a documented history of people being coerced into medical studies amid the Chinese Communist Party’s genocide targeting Uyghur Muslims and other minorities,” the select committee’s chairman John Moolenaar (R-MI) in a March 18 hearing on the generic drug supply chain.
“In spite of this forced testing, the FDA currently allows companies to use data from uninspected Chinese trial sites to advance drug applications in the United States,” he added.
The medical industry has its own set of ethical standards to regulate human testing. The International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use, which China joined in 2017, coordinates common safety and quality standards for drug development. In 2023, China’s State Council approved guidelines for ethical review committees, informed consent, and patient privacy protection in human clinical trials.
None of the companies referred to in this story have been accused of wrongdoing.

But Xinjiang is still a region where informed consent is difficult to obtain in an ethical way, says Darren Byler, assistant professor of International Studies at Canada’s Simon Fraser University, who focuses on Uyghur issues. “[The companies] are opening themselves up to some very serious concerns when it comes to human rights abuse.”
In the context of existing U.S. regulations designed to ensure supply chains are free from forced labor, coercion, and involvement with foreign militaries, clinical trial data and hospital partnerships are still a blind spot. Data from WireScreen shows that PLA hospitals are not sanctioned or included on the BIS Entity List or Section 1260H of the National Defense Authorization Act.
Existing U.S. Regulations Addressing PLA and Xinjiang Supply Chains
| Name | Enforced from | Covers | Applies to clinical trials | Applies to PLA hospitals |
|---|---|---|---|---|
| Comprehensive Outbound Investment National Security Act of 2025 | 2025 | Outbound investment restrictions: advanced tech, AI, chips | No | No |
| BIOSECURE Act (Section 851 of 2026 NDAA) | 2025 | Contracting with biotechnology companies of concern | No | No |
| Uyghur Forced Labor Prevention Act | 2022 | Import ban — Xinjiang goods | No | No |
| National Defense Authorization Act — Section 1260H | 2021 | Chinese military company restrictions | No | No |
| BIS Entity List | 2019 | Export license requirements — Xinjiang & military entities | No | No |
| NDAA Section 889 | 2018 | Chinese telecommunication companies | No | No |
Source: Congress, Federal Register
Existing FDA and U.S. Department of Health and Human Services regulations allow the FDA to investigate trial sites if necessary, but the questions posed by the Select Committee two years ago about whether these investigations were attempted in Xinjiang or at military hospitals remain unanswered.
“There’s a lot of opacity in much of the global supply chain in a lot of different industries,” says Byler. “It’s really only when these multinational corporations feel public pressure and pressure from government bodies that they begin to change marketing strategies or production strategies.”

Savannah Billman is a Staff Writer for The Wire China based in NYC. She previously worked at the National Committee on U.S.-China Relations.



