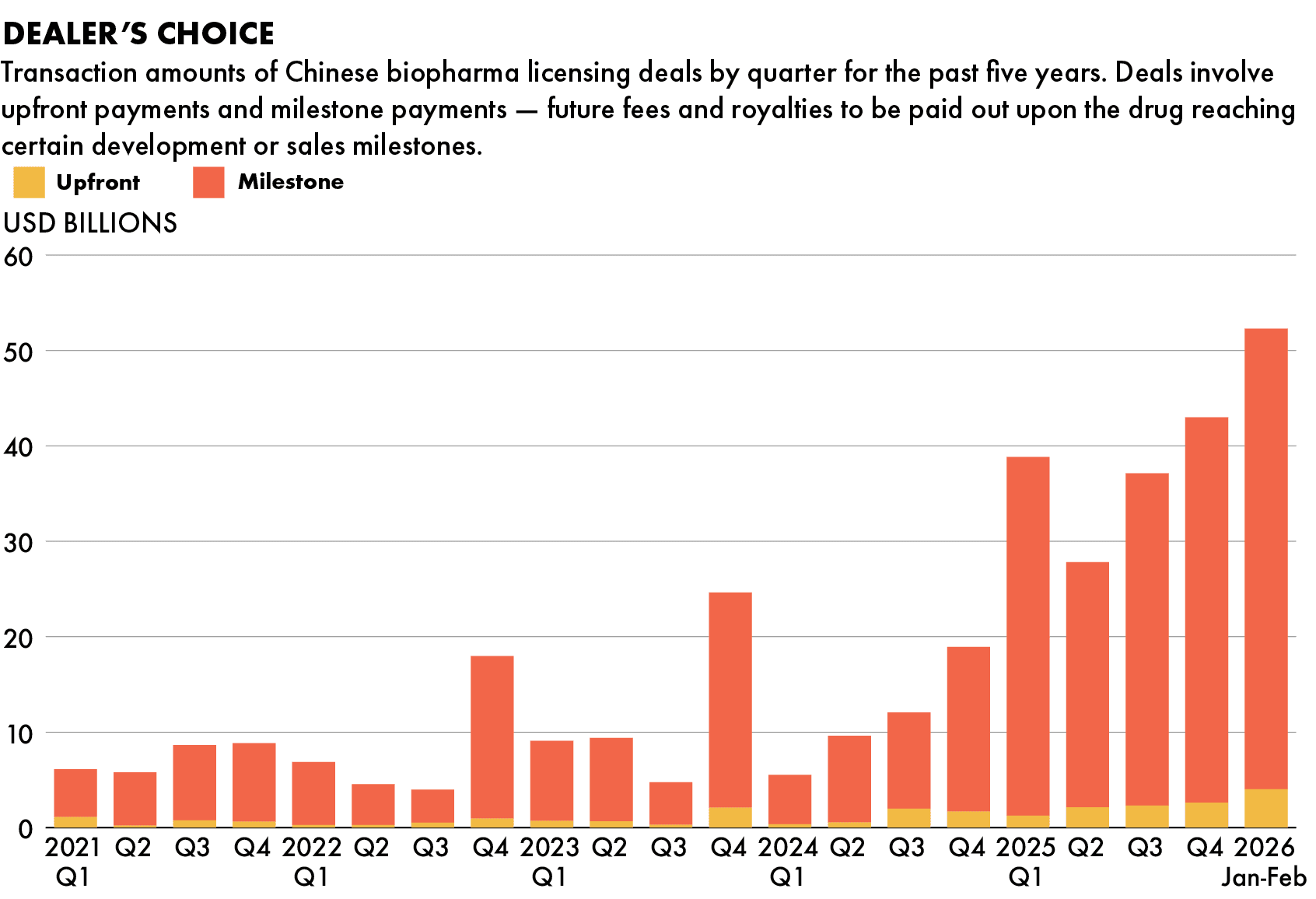
China’s biopharma industry is on a hot streak this year that not even geopolitical tensions can dull. Over $50 billion worth of licensing deals between Chinese and multinational firms were signed in the first two months of 2026 alone, a five-year high for quarterly deals.
Amid the flurry of licensing deals this year, two are notable for what they don’t include. Instead of purchasing rights to a specific existing treatment, the U.S.’s Eli Lilly and British company AstraZeneca licensed rights to unnamed drugs that, in some cases, have not yet been created.

Eli Lilly’s seventh collaboration with China’s Innovent Biologics, announced February 8, involves an upfront payment of $350 million for an undisclosed number of drugs that Innovent will develop domestically from concept to so-called ‘phase two’ clinical trials. Licensing deals between pharmaceutical companies typically involve specific drugs that are already in development.
“This is very different from your traditional license deal, because there’s no experimental drugs bought by Eli Lilly,” says Leon Tang, founder of the consulting firm InScienceWeTrust BioAdvisory.
AstraZeneca’s deal with CSPC, announced at the end of January, covers up to eight drug development programs, only four of which are in progress. Of these four, three programs have not yet progressed to human trials. The collaboration includes access to some of CSPC’s proprietary technologies, including an AI drug discovery platform.

“If you look at [the AstraZeneca-CSPC] deal, it has the same flavor [as Eli Lilly’s],” he adds. “By the time they closed the deal, that asset was not even in the clinic yet. That’s the crazy part.”
The uptick in dealmaking with Chinese firms comes despite regulatory pressure from the United States. The National Defense Authorization Act of 2026, signed into law in December 2025, included a revised version of the Biosecure Act, which prohibits U.S. companies from signing contracts with biotechnology companies operating on behalf of foreign adversary governments. In June 2025, the U.S. Food and Drug Administration halted clinical trials involving exports of Americans’ cells to labs in China and other ‘hostile countries.’

“If we’re seeing companies like AstraZeneca, Pfizer and Eli Lilly sign, it shows they’re all willing to take on that risk, despite any geopolitical risks like potential executive orders or the U.S. Biosecure Act,” says Dr. Ruby Wang, Director at LINTRIS Health consultancy and Editor at ChinaHealthPulse. “The quality [of the drugs] is too good.”
The close collaboration between global and Chinese biopharma firms runs counter to decoupling trends in other industries such as tech.
“We should be looking at industry actors and researchers because they’re the true technical experts in this space, and they’ll go wherever the good science is, and much of it is now all coming from China.” says Dr. Wang.
China Out-Licensing Deals Greater Than $100 Million from January and February
| Licensor | Licensee | Total Value |
|---|---|---|
| Innovent Biologics | 🇺🇸 Eli Lilly | $8.85 billion |
| Sanegene Bio | 🇨🇭 Genentech/Roche | $1.5 billion |
| Solstice Oncology | 🇨🇳 Harbour BioMed | $1.2 billion |
| CSPC Pharmaceuticals | 🇬🇧 AstraZeneca | $18.5 billion |
| RemeGen | 🇺🇸 Abbvie | $5.6 billion |
| Medilink Therapeutics | 🇨🇭 Roche | Undisclosed |
| SciNeuro | 🇨🇭 Novartis | $1.67 billion |
| Earendil Labs | 🇫🇷 Sanofi | $2.6 billion |
| Haisco Pharmaceutical | 🇺🇸 AirNexis Therapeutics | $1.06 billion |
| Structure Therapeutics | 🇨🇭 Roche | Undisclosed |
Source: PharmaDJ
TRIALS AND ERRORS
A familiar mix of government support, skilled talent, and manufacturing dominance has propelled China’s pharmaceutical industry to competitive levels, as it has done for other innovative industries.
“For people in the know, the investors and biotech heads who have been working on this for many years, it’s been an upwards trajectory, and what the rest of the world is finally now seeing — it was all sort of inevitable,” says Dr. Wang. The result is early-stage research and clinical trials involving human patients that are much cheaper in China than other major markets.

“In the U.S., if you want to do a gene therapy clinical trial for humans, typically it’s going to take you $20-25 million, [and] at least three years, to move from an idea to trying it on humans,” says Tang. In China, he says, the same trial might cost as little as $3-5 million and take as short as one year.
The timeline from early drug discovery to approval for human trials is also much quicker: 50 to 70 percent faster in China than the rest of the world, according to research by consultancy McKinsey.
China surpassed Europe in the number of clinical trials started in 2024, research from L.E.K. Consulting shows, though it still lags the United States. Out of 668 phase three trials carried out in China that year — the final testing stage with up to hundreds of human patients trialling the drug — one third were sponsored by multinationals, according to the same report.
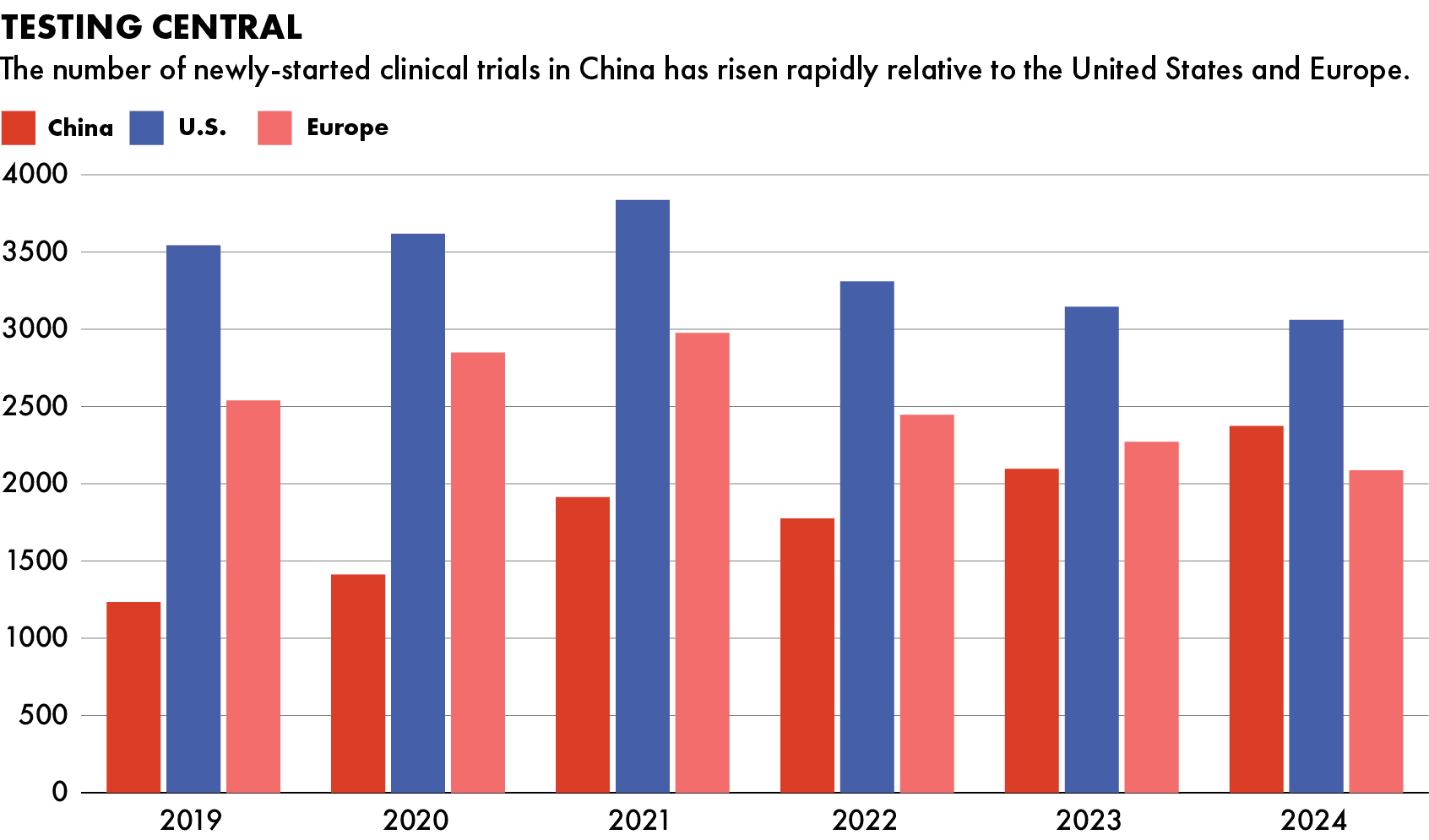
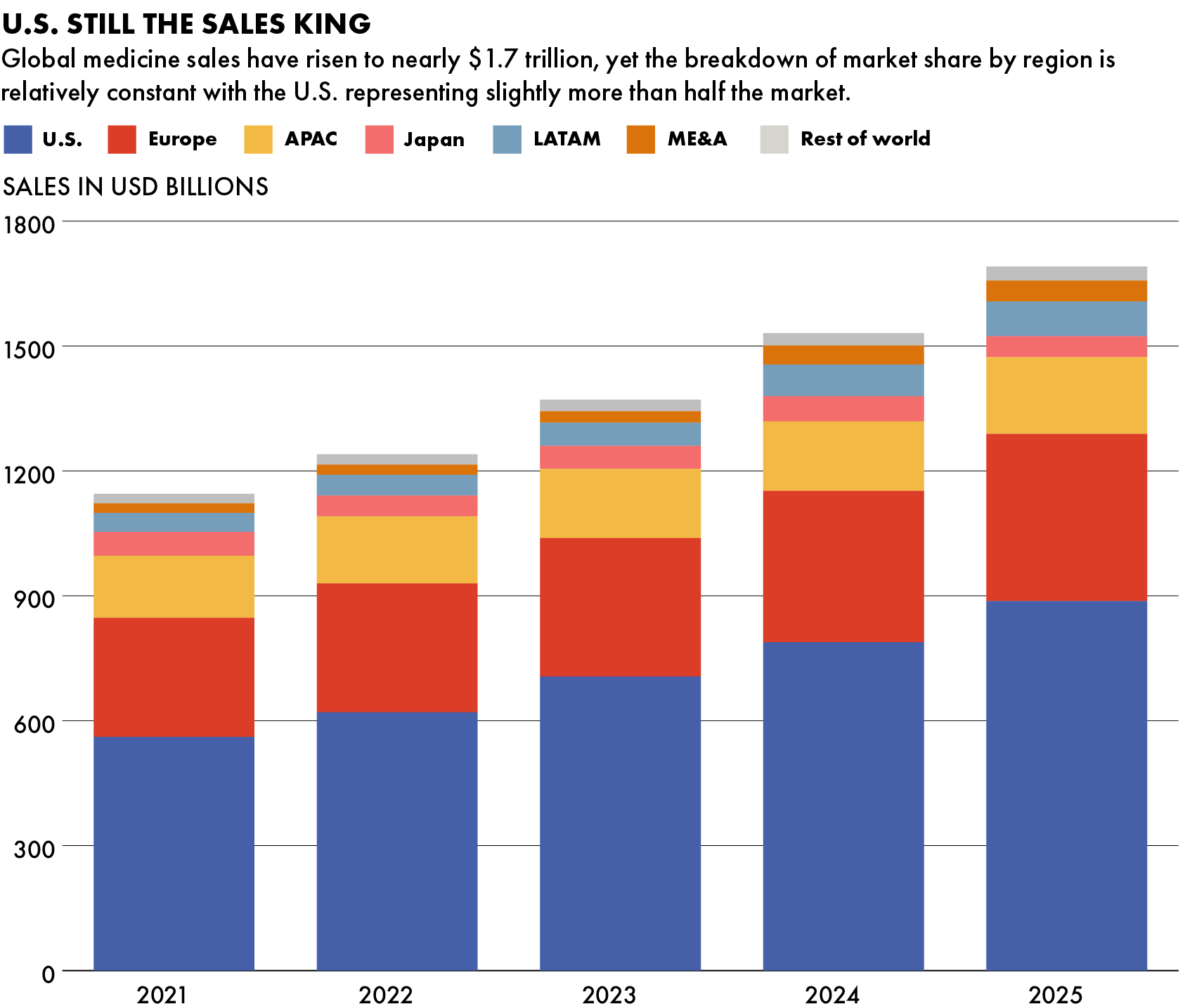
China’s R&D landscape is developing quickly, but multinationals still dominate in commercializing the drugs for profitable western markets.
Europe and the United States are the most profitable regions, with a combined three-quarters share of global sales of prescription medicines. The entire APAC region represents just 11 percent of all global sales, according to IQVIA, a U.S. life sciences data provider.
The same top-down planning that has helped grow China’s biopharma industry is hampering its profitability. Companies’ ability to bring new treatments to market in China is limited by the National Reimbursed Drugs List, a set of medications reimbursed by the country’s Basic Medical Insurance, which covers 95 percent of the population.
Getting a drug listed makes it immediately affordable to most of China’s population. But a drug or treatment’s inclusion on the NRDL comes with strict price caps. Companies looking to sell in China have to make tough choices between access and profit.

“There’s a paradox between health policy versus industrial strategy in China right now,” says Dr. Wang. “These biotechs are able to grow and deliver excellent medicines, but then they can’t sustain growth because they can’t sell those at high enough prices to make profit.”
So while China may be the best place for research and trials, for its companies to be profitable means collaborating with multinationals to commercialize their products overseas.
“It doesn’t matter how strong your biotech is,” Tang says. “In the end, big pharma is the customer of biotech companies.”

Savannah Billman is a Staff Writer for The Wire China based in NYC. She previously worked at the National Committee on U.S.-China Relations.



