
Much as in the world of artificial intelligence, 2025 has begun on a high note for Chinese companies in biotech, with global interest in their work never greater.

But unlike in AI, where U.S. players are feeling the heat and seeking to block Chinese competitors from the market, Chinese biotech companies and their international counterparts have found ways to collaborate so both sides can benefit. Two months into the year, the amount foreign pharmaceuticals have spent on acquiring Chinese drugs has already reached an all-time high.
Chengdu-based Keymed Biosciences struck two deals in January alone, selling the global rights to its early-stage drugs for auto-immune diseases to two American start-ups for $47.5 million in cash upfront, plus minority stakes in the acquiring firms. The same month, Innovent from Suzhou in Jiangsu province signed an agreement with the Swiss pharma giant Roche to jointly develop its lung cancer drug for $80 million immediately.
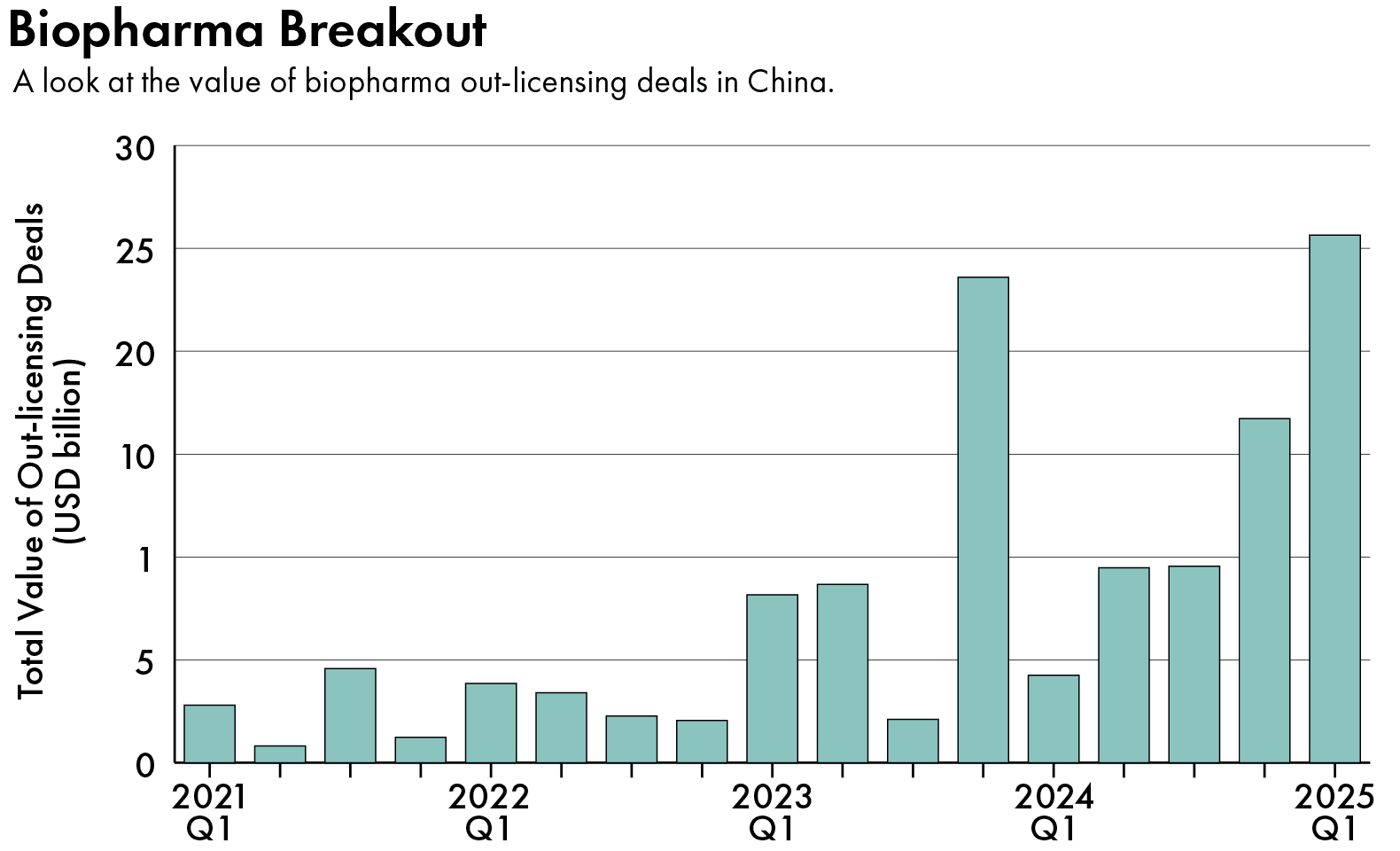
Such deals have helped raise China’s profile as a biotech innovation powerhouse at several recent industry conferences. The question is whether such partnerships can continue to thrive under the Trump administration, which has vowed to scrutinize Chinese investments in both healthcare and biotech.
In the old days, Chinese biotech firms had a lot of funding. They didn’t want to license to the big pharmas. They wanted to be the next big pharma. But right now it’s not easy for them.
Jialing Dai, founder of PharmaDJ, an industry intelligence firm
“The biotech industry as a whole is facing an unprecedented level of uncertainty that’s connected to geopolitical tension,” says Xiaoqing Boynton, a partner at DGA-Albright Stonebridge Group, where she focuses on healthcare and life sciences. “It needs to understand how that uncertainty is going to impact everything from the flow of data to the funding that’s needed for these kinds of licensing deals.”

Several reasons lie behind the growing interest in the molecules cooking up in Chinese labs. Chinese biotech firms are no longer making only incremental improvements to existing drugs; they are also discovering novel therapies — at a faster and cheaper rate than their international peers. China has also cut red tape so that clinical trials can proceed more swiftly.
At the same time, Chinese firms are having to deal with a capital-raising winter and thinning profit margins at home.
“In the old days, Chinese biotech firms had a lot of funding,” says Jialing Dai, founder of PharmaDJ, an industry intelligence firm. “They didn’t want to license to the big pharmas. They wanted to be the next big pharma. But right now it’s not easy for them.”

To scale up globally, Chinese companies would typically have to pour billions of dollars into clinical trials overseas, navigate new regulatory complexities, and build their own distributing network — spending which could go down the drain if their drugs were eventually rejected by foreign regulators, Dai adds.
So instead, many Chinese biotech firms have been turning to the safer option of out-licensing their products. That involves selling the rights to develop and commercialize their drugs in foreign markets to overseas partners, in return for immediate cash and future royalties.
The buyers are often multinational pharmaceutical companies, like Roche, on the hunt for the next blockbuster drug. For them, dealmaking in China can work well financially: upfront payments for in-licensing deals were on average nearly half the level paid for similar transactions in the U.S. last year, according to a report by investment bank Locust Walk.
“Such deals have become one of few sources of cash for Chinese biotech companies, especially when they have trouble raising funds,” says Alex Wang, founder of healthcare consultancy PJ Partner.
While Chinese biotech companies raised a total of $3.3 billion in fresh capital last year, they signed $40 billion worth of out-licensing deals, pocketing $4.2 billion in upfront payments, according to data firm PharmaDJ.
Chinese companies have in turn become crucial to the global pharmaceutical industry. About a third of the molecules acquired by multinational pharmaceutical companies last year came from Chinese companies, up from zero in 2019, data from DealForma shows.
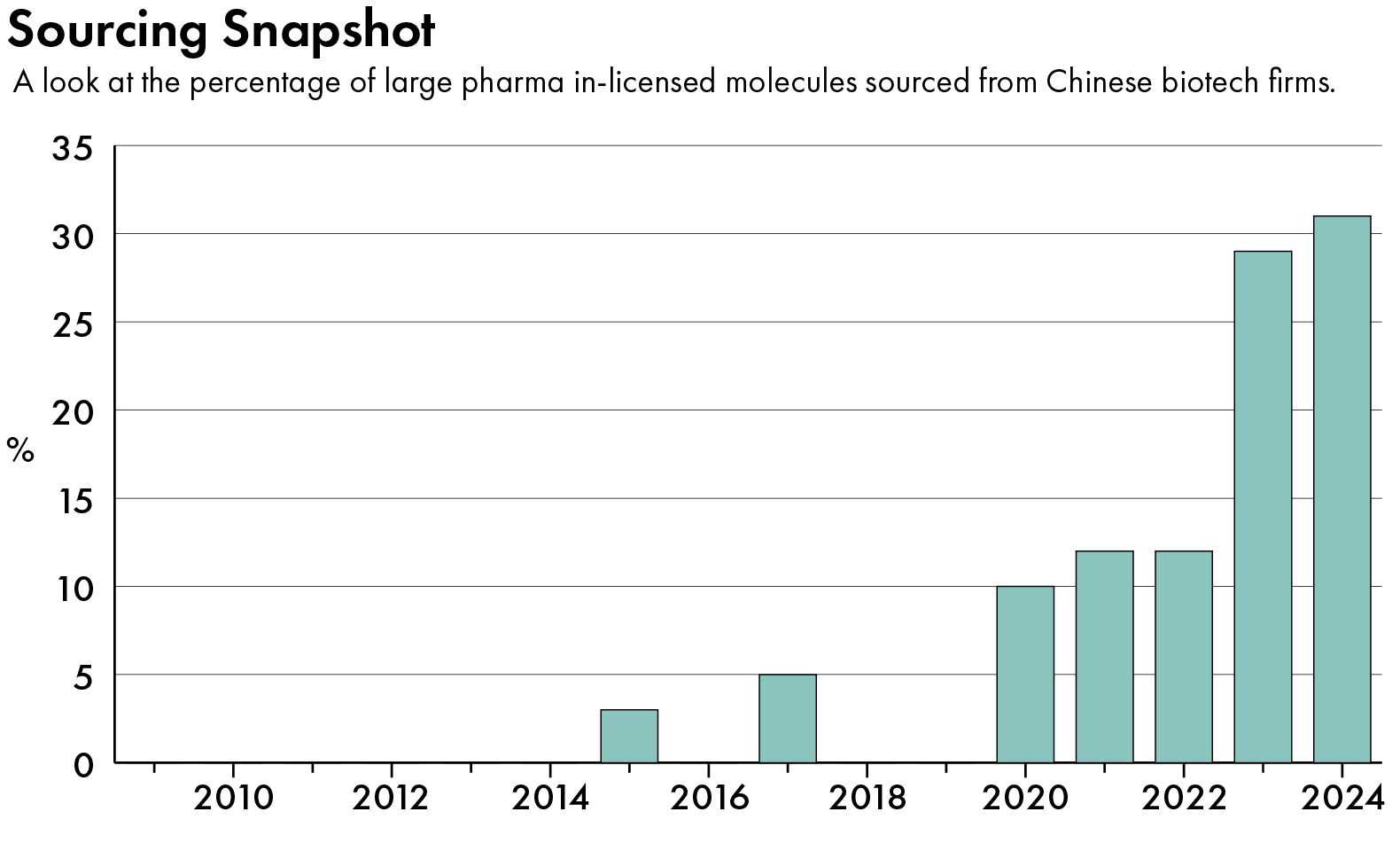
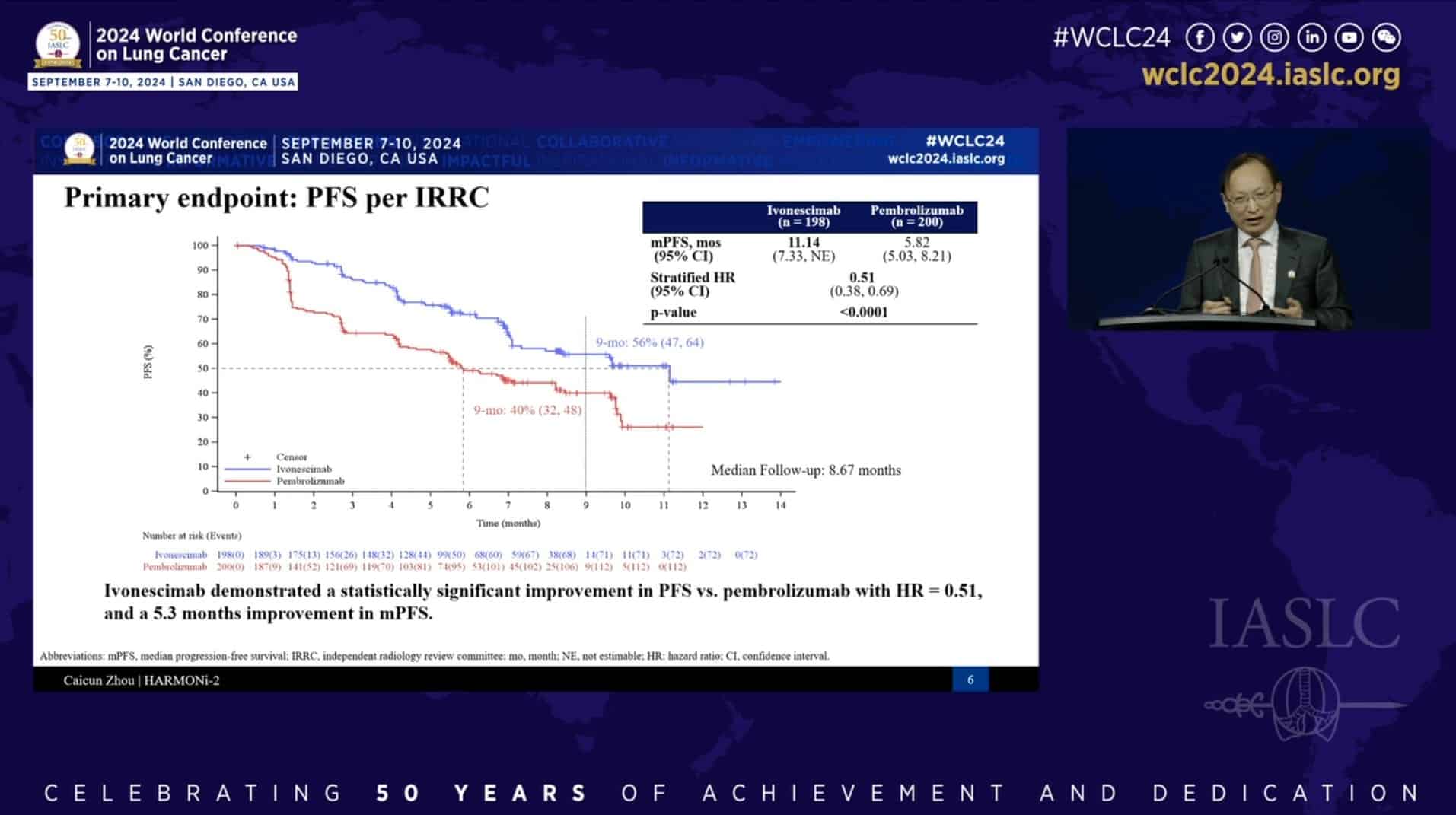
The Chinese industry chalked up another milestone last September, when a cancer drug developed by Shanghai-based Akeso outperformed Merck’s blockbuster drug Keytruda in a Chinese trial, cutting the risk of death by half. Last month, the company said the drug is proceeding with a global clinical trial overseen by its licensee, U.S. biotech firm Summit Therapeutics, and American pharmaceuticals giant Pfizer.
For Chinese companies that have yet to find a buyer, another option is to create a foreign spinoff — a so-called ‘NewCo’ — in which they hold a stake, with backing from foreign venture capital firms and run by independent management teams.
“As a U.S. company, [the new entity] would have more liberty and latitude to make deals, to go for an initial public offering or pursue other strategic options,” says Bruce Liu, a senior partner at the consultancy Simon-Kucher. “It’s a smart way to de-risk.”

Jiangsu Hengrui, a top pharmaceutical company in China, adopted this approach with the launch of Kailera Therapeutics in October, backed by Atlas Venture and Bain Capital Life, among other U.S. venture capital firms. Kailera acquired the global rights to Jiangsu Hengrui’s GLP-1 portfolio — the same type of medication as the weight-loss drug Ozempic. In return, Jiangsu Hengrui received $110 million upfront and could earn another $5.9 billion as the drugs move ahead.
GLP-1 is a drug with huge potential in many markets, says Su Zhang, former global head of business development at Jiangsu Hengrui who led the deal. “But taking that portfolio to do global development requires $2 billion. It’s not something a Chinese biotech or biopharma can take on.”
This whole industry is built upon low probability of success and the scarcity of asset innovation. All of a sudden, there’s this flurry of activity and availability of cheap innovation. I see new business models and consolidation one way or another.
Su Zhang, former global head of business development at Jiangsu Hengrui
Over a dozen such companies have been formed in the last two years, according to the market intelligence firm BioCentury, some involving assets from other parts of Asia. Keymed Biosciences, the firm from Chengdu, has set up four such entities.

Yet geopolitics is now casting a cloud over such initiatives. In a presidential memo last month, the Trump administration named healthcare as an area where the U.S. government will restrict investment from Chinese entities, and biotech as a sector where it will curb outbound investment.
The industry is watching to see how government agencies will implement these orders. But there are already concerns that the NewCo model will not stand. The memo, for instance, noted that the U.S. will continue to “welcome and encourage passive investments.”
“That’s going to pose a potential challenge for NewCo because the idea of Chinese intellectual property holders having equity, voting rights, board seats is the opposite of passive investment,” DGA-Albright Stonebridge Group’s Boynton says.
Restrictions on investments may also cut both ways. “This type of outlicensing deals provides a pathway to bring certain innovative assets to U.S. patients faster, and possibly through a more cost effective structure,” says Boynton.
In other words, it may be patients who suffer the consequences if the rules cut access to more affordable medicine.
No matter how the rules turn out, the sheer quantity and quality of drugs emerging from China will have wide-ranging implications, not least, for the tens of thousands of biotech firms in the country competing against each other.
“This whole industry is built upon low probability of success and the scarcity of asset innovation. All of a sudden, there’s this flurry of activity and availability of cheap innovation,” says Zhang. “I see new business models and consolidation one way or another. It’s not possible to have this many biotechs doing more or less similar things.”

Rachel Cheung is a staff writer for The Wire China based in Hong Kong. She previously worked at VICE World News and South China Morning Post, where she won a SOPA Award for Excellence in Arts and Culture Reporting. Her work has appeared in The Washington Post, Los Angeles Times, Columbia Journalism Review and The Atlantic, among other outlets.




